The foundation guide to body-safe piercing materials
Walk into a reputable piercing studio, and you will hear the words implant-grade more than almost any other term. It appears on jewellery packaging, in aftercare instructions, and in studio policies. It is used constantly, explained rarely, and misunderstood by the majority of people getting pierced.
That gap matters. Implant-grade is not a marketing phrase invented by jewellery brands to justify higher prices. It refers to specific, enforceable international standards for medical devices that are implanted in the human body.
The difference between jewellery that meets these standards and jewellery that does not is the difference between a piercing that heals cleanly and one that never quite settles.

This guide explains exactly what “implant-grade” means, which materials qualify and why, which materials do not qualify and why that matters, how to verify a piece of jewellery before you buy it, and what happens to piercings when the wrong material is used throughout the healing process.
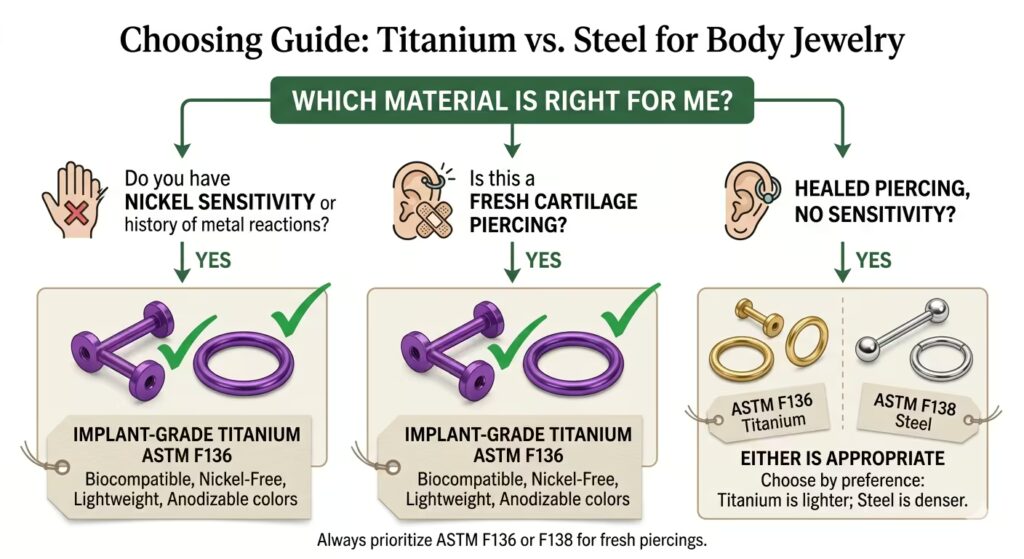
❝ Implant-grade is defined by specific ASTM International or ISO standard numbers. It is not defined by price, brand name, the words “surgical”, “hypoallergenic”, or “safe for sensitive skin” on a package. Always ask for the standard number.
What Implant-Grade Actually Means
The term implant-grade comes from the medical device industry. When a material is described as implant-grade, it means the material meets the standards set by ASTM International or the International Organization for Standardization (ISO) for devices designed to be placed inside the human body for extended periods.
These standards exist because the human body is extraordinarily sensitive to foreign materials. Metals, plastics, and ceramics that seem inert under normal environmental conditions can cause significant problems when placed in direct, continuous contact with living tissue.
The implant-grade standards define the specific composition, manufacturing processes, surface finish, and biocompatibility testing requirements that materials must meet to be considered safe for that contact.
A body piercing is, in practical terms, a medical implant situation. The jewellery sits within a channel of body tissue continuously for months during healing and potentially for years or decades afterward. The same safety logic that governs hip replacements, surgical staples, and bone plates applies to the piece of metal in a healing cartilage piercing.
The relevant standards for body piercing
The primary ASTM International standards referenced in professional body piercing are ASTM F136 for titanium, ASTM F138 for steel, and ASTM F2384 for niobium. The equivalent ISO standards are ISO 5832-3 for titanium and ISO 5832-1 for steel.
Any material claiming to be implant-grade for piercing use should be able to cite one of these specific standard numbers.
The Association of Professional Piercers, the primary professional body in the piercing industry, maintains a materials policy that specifies which standards are acceptable for initial piercing jewellery.
Their policy is publicly available and references these ASTM and ISO standards directly. A studio that follows APP guidelines will stock jewellery that meets these standards.

The Four Requirements Every Implant-Grade Material Meets
Understanding what specifically makes a material implant-grade helps you understand why some materials qualify and others do not. There are four distinct requirements that all implant-grade materials must satisfy.
1. Controlled alloy composition
Implant-grade standards specify the exact percentages of each element in the alloy and set maximum impurity limits. For ASTM F136 titanium, the standard specifies 5.5 to 6.5 percent aluminium, 3.5 to 4.5 percent vanadium, and strict maximum limits on iron, oxygen, nitrogen, carbon, and hydrogen content.
This precision matters because trace elements in an alloy can behave very differently from the same elements in isolation.
Nickel bound within an implant-grade steel alloy leaches at a far lower rate than free nickel in a cheaper alloy because the binding energy within the alloy matrix holds the nickel atoms more tightly. The composition standard controls exactly how the elements are combined.
2. Surface finish requirements
Implant-grade jewellery must be finished to a specific surface quality, typically a mirror polish achieved through mechanical polishing followed by electropolishing.
Electropolishing uses an electrolytic process to remove the outermost layer of metal, eliminating microscopic surface irregularities that are invisible to the naked eye but large enough to harbour bacteria and cause friction against healing tissue.
A rough or poorly finished surface on a piece of jewellery sitting in a healing piercing channel creates two problems simultaneously. It provides physical locations for bacteria to establish themselves that cannot be fully cleaned.
And the friction between a rough surface and the delicate cells forming the fistula wall continuously disrupts that formation. Surface finish is not cosmetic. It is clinical.
3. Biocompatibility testing
Materials intended for implant-grade designation are tested against the ISO 10993 standard for biological evaluation of medical devices. This testing evaluates cytotoxicity (whether the material kills cells), sensitisation (whether it causes allergic responses), irritation (whether it inflames tissue), and systemic toxicity (whether it causes broader harm to the body).
A material can look and feel inert and still fail biocompatibility testing. Certain plastics and metal alloys release compounds that disrupt cell function in ways that only become apparent under laboratory conditions. The biocompatibility testing requirement ensures that jewellery has been evaluated against these effects rather than simply assumed to be safe.
4. Corrosion resistance
Body tissue is a corrosive environment. Saliva, lymph fluid, blood, and sweat all contain salts and organic compounds that corrode metals that would otherwise seem stable. Implant-grade materials must demonstrate stability in this environment over extended periods.
Titanium forms a stable, passive oxide layer on its surface that resists corrosion exceptionally well. Implant-grade steel resists corrosion through its chromium content, which forms a protective chromium oxide layer. Materials that corrode in body tissue release breakdown products into the surrounding tissue, causing inflammation and potential toxicity.
Materials That Meet Implant-Grade Standards
| Material | Standard | Nickel-Free | Best Applications |
| Implant-Grade Titanium | ASTM F136 | Yes | All piercings at all stages. The current professional standard for initial jewellery. |
| Implant-Grade Steel (316LVM) | ASTM F138 | No (8–12% bound) | Healed piercings in non-sensitive individuals. Acceptable for initial by APP. |
| Niobium | ASTM F2384 | Yes | Excellent nickel-free alternative to titanium. Anodisable. Slightly heavier. |
| Solid 14k / 18k Gold (nickel-free alloy) | Nickel-free verified | Yes (yellow/rose) | Healed piercings. Premium long-term choice. Not ideal for fresh cartilage due to weight. |
| Medical-Grade PTFE / Bioplast | Medical grade | N/A (non-metallic) | Retainers, pregnancy belly bars, flexible applications. Not for decorative use. |
| Borosilicate Glass | Medical grade | N/A (non-metallic) | Stretched piercings, healed placements. Completely inert and smooth. |
Materials That Do Not Meet Implant-Grade Standards
Understanding what does not qualify as implant-grade is at least as important as understanding what does. The majority of body jewellery sold in high street stores, online marketplaces, and even some piercing studios does not meet implant-grade standards. Knowing how to identify these materials protects your piercing.
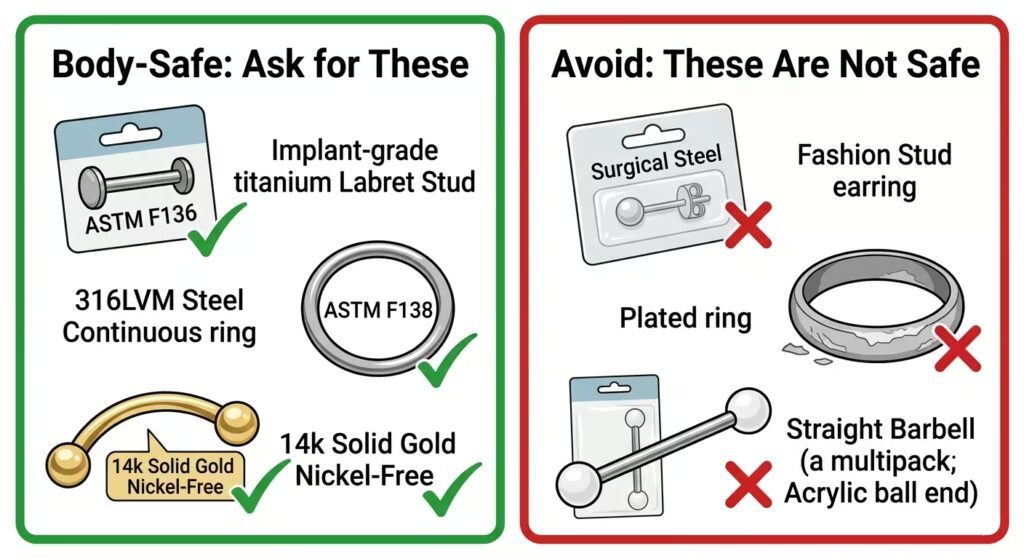
“Surgical steel” without a standard number
This is the most widespread and consequential labelling problem in body jewellery. The phrase “surgical steel” has no legal or regulatory definition in most countries. Any manufacturer can apply it to any stainless steel alloy regardless of its nickel content, surface finish, or actual biocompatibility.
Fashion jewellery labelled surgical steel is frequently made from 304-grade stainless steel. Grade 304 has a similar nickel content to 316L but exhibits a higher nickel leach rate in acidic and biological environments. In a healing piercing, this means significantly more nickel exposure to healing tissue than implant-grade 316LVM would produce.
The test is simple: ask for the ASTM or ISO standard number. If the answer is “surgical steel” or “hypoallergenic” without a number, the material is unverified. A reputable studio stocking genuine implant-grade steel will have 316LVM and ASTM F138 readily available to answer that question.
Gold-plated and gold-filled jewellery
Gold-plated jewellery has a thin layer of gold deposited on top of a base metal. The base metal in virtually all gold-plated fashion jewellery is a high-nickel alloy. The gold layer is thin and wears through with repeated insertion, removal, and contact with body fluids, exposing the nickel base metal directly to healing tissue.
Gold-filled jewellery has a thicker gold layer than plated jewellery, but still uses a base-metal core. The gold layer is mechanically bonded to the base metal rather than deposited. It is more durable than plated, but it is not body-safe for piercings because the base metal will eventually be exposed, and the bonding layer itself can cause irritation in sensitive tissue.
Neither gold-plated nor gold-filled jewellery should be used in any piercing at any stage. The only gold that is safe for piercings is solid gold with a verified nickel-free alloy composition. Our full guide to using gold jewelry in piercings covers the gold karat, alloy, and colour questions in complete detail.
Acrylic and generic plastic
Acrylic and generic plastic body jewellery are porous at a microscopic level. Even a piece of acrylic that looks smooth to the naked eye has surface irregularities large enough to harbour bacteria in quantities that cannot be eliminated by surface cleaning. Autoclaving, which is the standard sterilisation method for body jewellery, damages or destroys most acrylic materials.
Acrylic also releases small amounts of chemical compounds from its surface over time, a process called off-gassing. These compounds cause irritation and can trigger contact reactions in healing tissue.
Acrylic is never appropriate for any piercing at any stage of healing, including fully healed piercings, where it will sit against the skin for extended periods.
Sterling silver
Sterling silver is one of the most common misconceptions in body piercing. Silver is not a piercing material. Sterling silver is 92.5 percent silver and 7.5 percent copper. In the body tissue environment, sterling silver tarnishes.
The tarnish compounds, including silver sulphide and other silver compounds, can permanently stain the skin tissue around the piercing in a condition called argyria.
The copper content in sterling silver also causes irritation in healing tissue. Silver jewellery worn in piercings during healing is a consistent cause of dark discolouration around the piercing site that does not resolve when the jewellery is removed. Silver should never be used in any piercing, including long-established healed piercings.
“Hypoallergenic” labels without material specifications
Hypoallergenic has no regulatory definition in most countries. Manufacturers apply it freely to products that may still contain nickel, acrylic components, or unverified alloys. A product labelled hypoallergenic without a specific ASTM or ISO standard number is making a marketing claim rather than a material safety guarantee.
The correct question to ask about any jewellery presented as hypoallergenic is: “What is the specific material and what standard does it meet?” A material that is genuinely hypoallergenic in the clinical sense will have an answer to that question.
What Happens When the Wrong Material Is Used
Most piercing complications attributed to poor technique, improper placement, or inadequate aftercare are actually due to material issues. The jewellery is in contact with healing tissue twenty-four hours a day.
When that jewellery is leaching nickel, releasing acrylic compounds, or corroding in body fluids, the resulting tissue irritation is continuous and cannot be overcome with aftercare alone.
Persistent irritation bumps
Irritation bumps are the most common symptom of a material problem. They are small raised areas that form around one or both ends of the jewellery, caused by persistent low-level inflammation. When the jewellery material is the irritant, the bump will not resolve, no matter how carefully aftercare is followed, because the source of the inflammation is not being removed.
A piercing that develops an irritation bump within the first few weeks of getting a replacement piece of jewellery, even after previously healing well with different jewellery, is a strong indicator that the new jewellery material is the problem. Replacing it with verified implant-grade material usually resolves the bump within two to four weeks.
Contact dermatitis
Contact dermatitis from nickel presents as persistent redness, itching, and sometimes fluid-filled blisters around the piercing site. It is an immune response to nickel ions leaching from the jewellery into the surrounding tissue. The response does not resolve with aftercare because the antigen, the nickel, is continuously present.
Nickel contact dermatitis from piercing jewellery is one of the most common causes of sensitisation, which is the development of a nickel allergy where none existed before. Once sensitised, the reaction is typically permanent and extends beyond piercings to any nickel-containing object that comes into contact with the skin.
Skin staining from silver
Silver compounds deposited in skin tissue during tarnishing create a permanent grey-blue discolouration around the piercing site. This staining is typically irreversible.
It does not respond to topical treatment, does not fade significantly over time, and cannot be resolved by removing the jewellery. Sterling silver in a piercing is one of the few jewellery choices that can cause permanent cosmetic harm.
Persistent infections
Acrylic jewellery in particular harbours bacteria in its porous surface in quantities that cannot be eliminated by the body’s local immune response or by surface cleaning. A piercing that develops recurrent infections may not be due to an infection management problem.
It may have a problem with the jewellery material. Replacing acrylic with implant-grade material and treating the acute infection appropriately typically resolves the cycle.
❝ Most piercing complications that do not respond to improved aftercare are material problems. Changing the aftercare routine without changing the jewellery material will not resolve an irritation caused by the jewellery itself.
How to Verify Implant-Grade Jewellery
Verification of jewellery material is a practical process that involves asking specific questions and knowing which answers are acceptable. The following approach works whether you are booking at a studio or purchasing jewellery online.
In a studio
Ask your piercer: “What material is this jewellery and what ASTM or ISO standard does it meet?” A studio that stocks genuine implant-grade jewellery will answer this immediately and specifically. “ASTM F136 titanium from Anatometal” or “316LVM steel to ASTM F138 from Industrial Strength” are the kinds of answers that indicate responsible sourcing.
If the answer is “surgical steel” or “it’s hypoallergenic” without a standard number, ask to see the supplier documentation. If that is unavailable, the jewellery material remains unverified. This is a reasonable question to ask, and a professional studio will not be offended by it.
Online purchasing
Look for the ASTM or ISO standard number in the product listing description. For titanium, ASTM F136 or “implant-grade ASTM F136 titanium” should appear in the listing. For steel, ASTM F138 and the 316LVM designation should be present. For gold, confirmation that the alloy is nickel-free and the karat designation (14k or 18k) should be clear.
Established body jewellery retailers whose material standards are well-documented include Anatometal, Neometal, Industrial Strength, BVLA, Invictus, Junipurr Jewelry, and LeRoi.
Purchasing from these suppliers provides a high degree of material certainty. Purchasing from general online marketplaces without specific standard documentation carries significant uncertainty about actual material composition.
Price as a rough guide
Genuine implant-grade titanium from a certified supplier starts around $15 to $20 for a basic flat-back labret stud. Implant-grade steel starts around $8 to $12. Solid 14k gold body jewellery starts around $55 to $80 for a simple piece.
These are floors, not ceilings. Decorative pieces, gem-set pieces, and pieces from premium designers cost considerably more. But anything significantly below these price points claiming to be implant-grade warrants careful scrutiny. The raw material and manufacturing costs of genuine implant-grade production set a minimum price that very cheap jewellery cannot meet.

Implant-Grade Standards by Material: The Full Reference
Titanium: ASTM F136 / ISO 5832-3
The standard specifies Ti6Al4V ELI alloy: 5.5 to 6.5 percent aluminium, 3.5 to 4.5 percent vanadium, with strict maximum impurity levels for oxygen (0.13%), nitrogen (0.05%), carbon (0.08%), hydrogen (0.012%), and iron (0.25%). The ELI designation (Extra Low Interstitials) controls these impurity levels below standard titanium alloy thresholds.
The surface finish requirement specifies a mechanically polished surface, then electropolished. Anodised titanium jewellery can meet this standard when the base material and initial surface preparation are to ASTM F136 specification, and the anodising is performed as a subsequent step without introducing coatings or dyes.
Steel: ASTM F138 / ISO 5832-1
The standard specifies 316L stainless steel composition: 16 to 18 percent chromium, 10 to 14 percent nickel, 2 to 3 percent molybdenum, with maximum limits on carbon (0.03%), phosphorus (0.025%), and sulphur (0.01%).
The LVM designation requires the steel to be produced by vacuum induction melting followed by vacuum arc remelting, which eliminates inclusions and non-metallic impurities.
The low carbon content (L designation) and the controlled nickel percentage, combined with the LVM manufacturing process, produce a steel that resists corrosion and maintains nickel in a tightly bound alloy state with a very low leach rate. This is fundamentally different from the generic 304-grade steel used in fashion jewellery.
Niobium: ASTM F2384
Niobium is a pure element rather than an alloy. ASTM F2384 specifies minimum niobium purity, surface finish requirements, and mechanical properties for surgical implant use. Niobium is completely nickel-free and shares titanium’s ability to be anodised for colour.
It is slightly heavier than titanium and slightly less widely available, but is an excellent option for individuals who want a nickel-free alternative with colour options. Its use in body piercing is less universal than titanium, but it is fully accepted as an implant-grade material by the APP.
Gold: Nickel-free alloy verification
Gold does not have an ASTM implant-grade standard, as metallic alloys do. Instead, the safety verification for gold in piercings is the karat designation (14k minimum for durability), the alloy composition (nickel-free), and the piece’s manufacturing quality.
Yellow gold and rose gold alloys are typically nickel-free. White gold must be specifically confirmed as a palladium alloy rather than traditional nickel-based white gold.
For the complete guide to gold in piercings, including karat explanations, colour safety, and when gold is and is not appropriate, see our dedicated article on using gold jewelry in piercings.
The Sterilisation Question
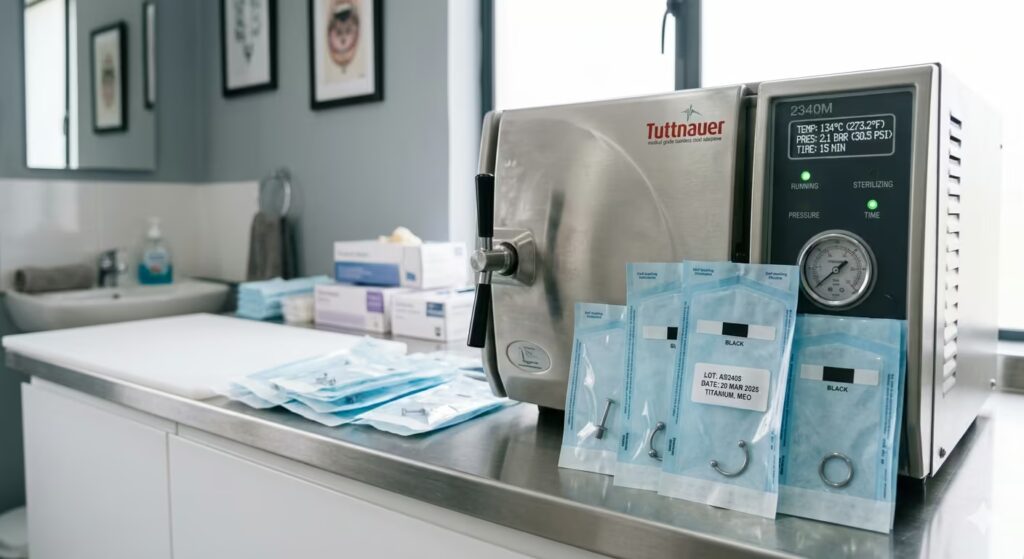
Implant-grade jewellery certification applies to the material composition and surface finish of the jewellery. It does not automatically mean the piece is sterile when you receive it or when it is inserted into a piercing. Sterilisation is a separate step that must be performed correctly for the jewellery to be safely introduced to a piercing environment.
Professional piercing studios sterilise jewellery in an autoclave before insertion. An autoclave uses pressurised steam at high temperature to kill all microorganisms, including spores. Properly run autoclaves with validated cycles are the gold standard for jewellery sterilisation.
Jewellery purchased online and inserted into a piercing without autoclave sterilisation may be made from implant-grade material but will not be sterile. For fresh piercings, this distinction matters.
For jewellery changes in established, healed piercings, the risk is lower, but cleaning the new jewellery thoroughly before insertion remains good practice.
Implant-Grade Jewellery for Each Piercing Stage
The material requirements for jewellery do not change between fresh and healed piercings in terms of which materials are body-safe. But the specific priorities within those requirements shift as a piercing matures.
Fresh piercings: the strictest requirements
During the healing period, the piercing channel is essentially an open wound in continuous contact with the jewellery. Any material deficiency, any nickel leaching, any surface roughness, any chemical off-gassing, is experienced by healing tissue that has no defensive barrier against it.
For fresh piercings, the recommendation is ASTM F136 implant-grade titanium as the default. It is the material that creates the fewest possible complications for the widest range of people.
Implant-grade steel ASTM F138 is the accepted alternative for non-sensitive individuals. No other metallic material meets the standard for fresh piercing jewellery in professional practice.
Healed piercings: more flexibility, same principles
A fully healed fistula has a stable epithelial lining that provides a barrier between the jewellery and the underlying tissue. This means healed piercings are less vulnerable to material problems than fresh ones, and the range of body-safe options expands to include solid gold, niobium, and high-quality implant-grade steel for people who did not use them during healing.
The core principle remains: the material must be body-safe, verified, and non-reactive. The key change is that the consequence of a material compromise develops more slowly in healed tissue than in healing tissue, giving the body more time to manage minor irritation before it becomes a significant problem.
For comprehensive guidance on when to change jewellery at different healing stages, our article on when to change my ear piercing covers the full healing timeline breakdown and what each stage means for jewellery decisions.
Frequently Asked Questions
Is all titanium body jewellery implant-grade?
No. Titanium is a broad category of metals with many different alloy compositions used across industrial, aerospace, consumer electronics, and medical applications. Only titanium that meets ASTM F136 or ISO 5832-3 is implant-grade.
“Titanium” on a jewellery listing without a standard number is unverified and could be any of a wide range of titanium alloys with varying biocompatibility.
Why does my studio charge more for implant-grade jewellery?
Genuine implant-grade jewellery is more expensive to produce than fashion jewellery. The raw materials are more expensive, the manufacturing processes are more controlled, the surface finishing is more labour-intensive, and the certification and documentation add to the supply chain cost. A studio charging more for implant-grade jewellery is passing on the real cost of genuine material quality rather than marking up a cheap product.
Can I bring my own jewellery to be pierced with?
Many studios accept client-supplied jewellery if it meets their material standards and can be verified. The jewellery will need to be autoclaved before insertion. Some studios charge for this service. Bringing jewellery from a reputable body jewellery supplier with clear documentation of materials gives it the best chance of being accepted.
Bringing fashion jewellery, mystery metal, or unverified pieces is likely to result in the studio declining to use them. This is not an arbitrary policy. It is a professional standard that protects the client. A studio willing to pierce any jewellery a client brings, regardless of material, is not adhering to appropriate professional standards.
How do I store implant-grade jewellery safely?
Store implant-grade jewellery in a clean, closed container away from harsh chemicals, bleach, and direct sunlight. Titanium with anodised colour can fade slowly with exposure to strong UV light. Steel should be kept away from prolonged moisture to prevent surface marks, though genuine 316LVM steel is highly corrosion-resistant under normal storage conditions.
Jewellery that has been stored for a long time should be cleaned and, ideally, autoclaved before reinsertion into a piercing, even a fully healed one. Dust and airborne contamination on stored jewellery can introduce bacteria into the piercing channel.
Is implant-grade jewellery MRI-safe?
Implant-grade titanium and niobium are non-ferromagnetic materials, meaning they are not attracted to magnetic fields and do not create significant artefacts in MRI imaging. They are generally considered MRI-safe. Implant-grade steel, while not strongly magnetic, can sometimes create minor imaging artefacts depending on the specific MRI protocol and the location of the jewellery.
Always inform your MRI technician about any body jewellery before a scan. In most cases, jewellery should be removed if possible. If a piercing cannot be safely removed, your technician can advise whether it poses any issues for the specific scan being performed.
What is the difference between implant-grade and body-safe?
Implant-grade refers specifically to materials meeting ASTM or ISO standards for surgical implants. Body-safe is a broader term that includes implant-grade materials but also other materials, like borosilicate glass, known to be safe for body tissue contact without a specific surgical implant standard.
In piercing contexts, the terms are often used interchangeably, but implant-grade is the more specific and verifiable claim. When a studio says “body-safe” without a standard number, it is worth clarifying exactly what they mean.
The Material Is the Foundation
Everything else in piercing aftercare, every cleaning routine, every sleeping position adjustment, every dietary change during healing, sits on top of the foundation of the jewellery material. If that foundation is wrong, the other elements of aftercare cannot compensate for it.
Implant-grade jewellery is not a premium option for those seeking the best experience. It is the baseline standard for safe piercing. The standards exist because decades of medical research have established precisely which materials the human body tolerates and which it reacts to.
Working with those standards rather than against them is the most fundamental and most impactful decision in any piercing journey.
Ask for the standard number. Verify the supplier. Choose titanium for fresh piercings and cartilage. Graduate to gold for healed piercings if you prefer it. And never use anything that cannot answer the question of what it is made from and to what standard. That verification is what separates a body-safe choice from a marketing claim, and a well-healed piercing from a persistent problem.